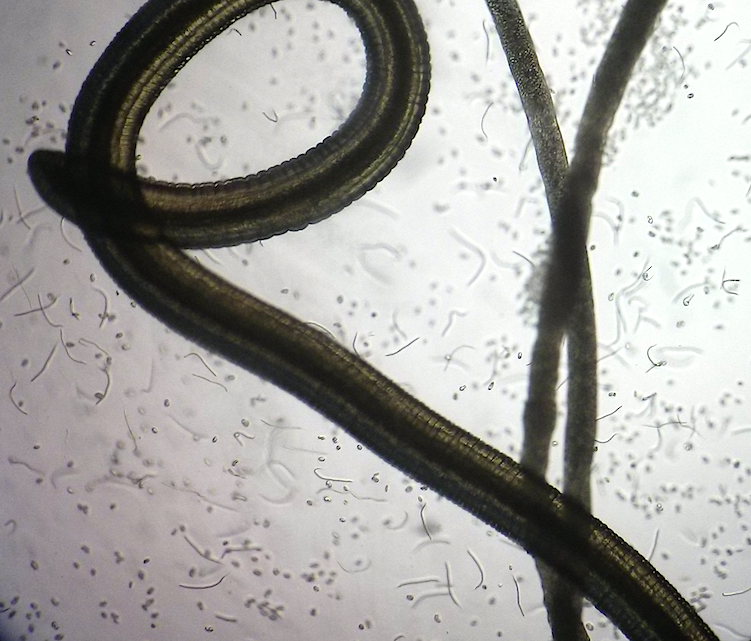
Researchers at the LSTM’s Centre for Drugs and Diagnostics (CDDR), and University of Buea, Cameroon, have developed new models of the tropical eye worm, Loa loa, for the development of new drugs against filariasis.
The research, led by Dr Joseph Turner at LSTM’s Department of Tropical Disease Biology, in collaboration with Professor Samuel Wanji’s team at The University of Buea, Cameroon, was published in Nature Communications.
Co-infection with Loa loa causes loiasis, which is a major barrier to the elimination of onchocerciasis (river blindness) a major neglected tropical disease (NTD). It is intended that the new models will accelerate the development of urgently needed alternative interventions, including novel therapies which are safe in loiasis patients, to accelerate elimination of river blindness in Central Africa.
“The current river blindness elimination strategy uses annual mass administration of ivermectin to communities where onchocerciasis infection is present” Dr Turner explained. “People who harbour high levels of L. loa are at risk of developing severe adverse reactions to ivermectin, which can cause coma and death. Therefore, many pharmaceutical and academic groups are developing new therapies which hope to target onchocerciasis whilst avoiding loiasis adverse reactions. However, a lack of accurate laboratory models of loiasis has hindered evidence-based decision-making of which drug candidates to progress. The key advance in our study was to utilise specific immunodeficient mouse strains as successful L. loa infection models which precisely emulate the clinical drug activity of ivermectin”.
Dr Turner continued: “This is a result of a longstanding and highly productive research collaboration with our colleagues at The University of Buea. The work required state-of-the art laboratory infrastructure to be established in Cameroon and extensive training of local graduate researchers. This scientific and capacity-strengthening research was only possible through grant funding from The Bill and Melinda Gates Foundation Grand Challenges Fund. We now believe the new facility is unique within Central Africa. It is gratifying that the new loiaisis preclinical testing models are operational within the disease endemic setting and with our support are being run on the ground by a Cameroonian scientific team. Thus far, we have been able to deploy the research model to scrutinise safety of 14 unique Onchocerca drug candidates, including regimens from our anti-Wolbachia (A-WOL) consortium. The model has proved invaluable in the prioritisation of moving drug candidates forward to preclinical development.”
Dr Turner and Professor Wanji’s team now hope that the novel research models can be used in a range of other translational medicine applications for tropical diseases that affect several hundred thousands of people in some of the most medically-disadvantaged communities of sub-Saharan Africa, including researching safe drug treatments specifically for loiasis, determining the mechanisms by which ivermectin causes adverse reactions in L. loa patients and validating new filarial diagnostics.
Mouse models of Loa loa
Nicolas P. Pionnier, Hanna Sjoberg, Valerine C. Chunda, Fanny F. Fombad, Patrick W. Chounna, Abdel J. Njouendou, Haelly M. Metuge, Bertrand L. Ndzeshang, Narcisse V. Gandjui, Desmond N. Akumtoh, Dizzle B. Tayong, Mark J. Taylor, Samuel Wanji & Joseph D. Turner. Nature Communicationsvolume 10, Article number: 1429 (2019)